The Herpesviridae are a large family of DNA viruses that cause diseases in animals, including humans. The members of this family are also known as herpesviruses. The family name is derived from the Greek word herpein ("to creep"), referring to the latent, recurring infections typical of this group of viruses. Herpesviridae can cause latent or lytic infections.
Viral structure
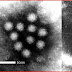
Herpesviruses all share a common structure—all herpesviruses are composed of relatively large double-stranded, linear DNA genomes encoding 100-200 genes encased within an icosahedral protein cage called the capsid which is itself wrapped in a protein layer called the tegument containing both viral proteins and viral mRNAs and a lipid bilayer membrane called the envelope. This whole particle is known as a virion.
Herpes virus life-cycle
All herpesviruses are nuclear-replicating—the viral DNA is transcribed to RNA within the infected cell's nucleus.
Infection is initiated when a viral particle contacts a cell with specific types of receptor molecules on the cell surface. Following binding of viral envelope glycoproteins to cell membrane receptors, the virion is internalized and dismantled, allowing viral DNA to migrate to the cell nucleus. Within the nucleus, replication of viral DNA and transcription of viral genes occurs.
During symptomatic infection, infected cells transcribe lytic viral genes. In some host cells, a small number of viral genes termed latency associated transcript (LAT) accumulate instead. In this fashion the virus can persist in the cell (and thus the host) indefinitely. While primary infection is often accompanied by a self-limited period of clinical illness, long-term latency is symptom-free.
Reactivation of latent viruses has been implicated in a number of diseases (e.g. Shingles, Pityriasis Rosea). Following activation, transcription of viral genes transitions from latency-associated LAT to multiple lytic genes; these lead to enhanced replication and virus production. Often, lytic activation leads to cell death. Clinically, lytic activation is often accompanied by emergence of non-specific symptoms such as low grade fever, headache, sore throat, malaise, and rash as well as clinical signs such as swollen or tender lymph nodes and immunological findings such as reduced levels of natural killer cells.
Taxonomy
The genus Herpesvirus was established in 1971 in the first report of the International Committee on Taxonomy of Viruses (ICTV). This genus consisted of 23 viruses and 4 groups of viruses. In the second ICTV report in 1976 this genus was elevated to family level - the Herpetoviridae. Because of possible confusion with viruses derived from reptiles this name was changed in the third report in 1979 to Herpesviridae. In this report the family Herpesviridae was divided into 3 subfamilies (Alphaherpesvirinae, Betaherpesvirinae and Gammaherpesvirinae) and 5 unnamed genera: 21 viruses were listed. In 2009 the family Herpesviridae was elevated to the order Herpesvirales. This elevation was necessitated by the discovery that the herpes viruses of fish and molluscs were only distantly related to those of birds and mammals. Two new families were created - the family Alloherpesviridae which incorporates bony fish and frog viruses and the family Malacoherpesviridae which contains those of molluscs.
This order currently has 3 families, 3 subfamilies plus 1 unassigned, 17 genera, 90 species and plus 48 as yet unassigned viruses.
Virus naming system
The system of naming herpes viruses was originated in 1973 and has been elaborated considerably since. The recommended naming system specified that each herpes virus should be named after the taxon (family or subfamily) to which its primary natural host belongs. The subfamily name is used for viruses from members of the family Bovidae or from primates (the virus name ending in –ine, e.g. bovine) and the host family name for other viruses (ending in –id, e.g. equid). Human herpes viruses have been treated as an exception (human rather than hominid). Following the host-derived term, the word herpes virus is added, followed by an arabic number (1,2,3,...). These last two additions bear no implied meaning about taxonomic or biological properties of the virus.
Some exceptions to this system exist. A number of viruses' names (e.g. Epstein–Barr virus) are so widely used that it is impractical to attempt to insist on their replacement. This has led to the a dual nomenclature in the literature for some herpes viruses. All herpes viruses described since this system was adopted have been named in accordance with it.
Immune system evasions
cmvIL-10
Herpesviruses are known for their ability to establish lifelong infections. One way this is possible is through immune evasion. Herpesviruses have found many different ways to evade the immune system. One such way is by encoding a protein mimicking human interleukin 10 (hIL-10) and another is by downregulation of the Major Histocompatibility Complex II (MHC II) in infected cells. Research conducted on cytomegalovirus (CMV) indicates that the viral human IL-10 homolog, cmvIL-10, is important in inhibiting pro-inflammatory cytokine synthesis. The cmvIL-10 protein has 27% identity with hIL-10 and only one conserved residue out of the nine amino acids that make up the functional site for cytokine synthesis inhibition on hIL-10. There is, however, much similarity in the functions of hIL-10 and cmvIL-10. Both have been shown to down regulate IFN-γ, IL-1α, GM-CSF, IL-6 and TNF- α, which are all pro-inflammatory cytokines. They have also been shown to play a role in downregulating MHC I and MHC II and up regulating HLA-G (non-classical MHC I). These two events allow for immune evasion by suppressing the cell-mediated immune response and natural killer cell response, respectively. The similarities between hIL-10 and cmvIL-10 may be explained by the fact that hIL-10 and cmvIL-10 both use the same cell surface receptor, the hIL-10 receptor. One difference in the function of hIL-10 and cmvIL-10 is that hIL-10 causes human peripheral blood mononuclear cells (PBMC) to both increase and decrease in proliferation whereas cmvIL-10 only causes a decrease in proliferation of PBMCs. This indicates that cmvIL-10 may lack the stimulatory effects that hIL-10 has on these cells.
It was found that cmvIL-10 functions through phosphorylation of the Stat3 protein. It was originally thought that this phosphorylation was a result of the JAK-STAT pathway. However, despite evidence that JAK does indeed phosphorylate Stat3, its inhibition has no significant influence on cytokine synthesis inhibition. Another protein, PI3K, was also found to phosphorylate Stat3. PI3K inhibition, unlike JAK inhibition, did have a significant impact on cytokine synthesis. The difference between PI3K and JAK in Stat3 phosphorylation is that PI3K phosphorylates Stat3 on the S727 residue whereas JAK phosphorylates Stat3 on the Y705 residue. This difference in phosphorylation positions seems to be the key factor in Stat3 activation leading to inhibition of pro-inflammatory cytokine synthesis. In fact, when a PI3K inhibitor is added to cells, the cytokine synthesis levels are significantly restored. The fact that cytokine levels are not completely restored indicates there is another pathway activated by cmvIL-10 that is inhibiting cytokine synthesis. The proposed mechanism is that cmvIL-10 activates PI3K which in turn activates PKB (Akt). PKB may then activate mTOR, which may target Stat3 for phosphorylation on the S727 residue.[6]
Another one of the many ways in which herpes viruses evade the immune system is by down regulation of MHC I and MHC II. This is observed in almost every human herpesvirus. Down regulation of MHC I and MHC II can come about by many different mechanisms, most causing the MHC to be absent from the cell surface. As discussed above, one way is by a viral chemokine homolog such as IL-10. Another mechanism to down regulate MHCs is to encode viral proteins that detain the newly formed MHC in the endoplasmic reticulum (ER). The MHC cannot reach the cell surface and therefore cannot activate the T cell response. The MHCs can also be targeted for destruction in the proteasome or lysosome. The ER protein TAP also plays a role in MHC down regulation. Viral proteins inhibit TAP preventing the MHC from picking up a viral antigen peptide. This prevents proper folding of the MHC and therefore the MHC does not reach the cell surface.
It is important to note that HLA-G is often up regulated in addition to downregulation of MHC I and MHC II. This prevents the natural killer cell response.[citation needed]
There are eight distinct viruses in this family known to cause disease in humans.[8][9]
Human Herpesvirus (HHV) classification
Type Synonym Subfamily Primary Target Cell Pathophysiology Site of Latency Means of Spread
HHV‑1 Herpes simplex virus-1 (HSV-1) α (Alpha) Mucoepithelial Oral and/or genital herpes (predominantly orofacial), as well as other herpes simplex infections Neuron Close contact (sexually transmitted disease)
HHV-2 Herpes simplex virus-2 (HSV-2) α Mucoepithelial Oral and/or genital herpes (predominantly genital), as well as other herpes simplex infections Neuron Close contact (sexually transmitted disease)
HHV-3 Varicella zoster virus (VZV) α Mucoepithelial Chickenpox and shingles Neuron Respiratory and close contact (including sexually transmitted disease)
HHV-4 Epstein-Barr virus (EBV), lymphocryptovirus γ (Gamma) B cells and epithelial cells Infectious mononucleosis, Burkitt's lymphoma, CNS lymphoma in AIDS patients,
post-transplant lymphoproliferative syndrome (PTLD), nasopharyngeal carcinoma, HIV-associated hairy leukoplakia B cell Close contact, transfusions, tissue transplant, and congenital
HHV-5 Cytomegalovirus (CMV) β (Beta) Monocyte, lymphocyte, and epithelial cells Infectious mononucleosis-like syndrome,[10] retinitis, etc. Monocyte, lymphocyte, and ? Saliva
HHV-6 Roseolovirus, Herpes lymphotropic virus β T cells and ? Sixth disease (roseola infantum or exanthem subitum) T cells and ? Respiratory and close contact?
HHV-7 Roseolovirus β T cells and ? Sixth disease (roseola infantum or exanthem subitum) T cells and ? ?
HHV-8 Kaposi's sarcoma-associated herpesvirus
(KSHV), a type of rhadinovirus γ Lymphocyte and other cells Kaposi's sarcoma, primary effusion lymphoma, some types of multicentric Castleman's disease B cell Close contact (sexual), saliva?
[edit] Herpesviruses of other animals
In addition to the herpesviruses considered endemic in humans, some viruses associated primarily with animals may infect humans. These are zoonotic infections:
Zoonotic Herpesviruses
Species Type Synonym Subfamily Human Pathophysiology
Macaque monkey CeHV-1 Cercopithecine herpesvirus-1, (Monkey B virus) α Very unusual, with only approximately 25 human cases reported.[11] Untreated infection is often deadly; sixteen of the 25 cases resulted in fatal encephalomyelitis. At least four cases resulted in survival with severe neurologic impairment.[11][12] Symptom awareness and early treatment are important for laboratory workers facing exposure.
Mouse MuHV‑4 Murine gammaherpesvirus-68 (MHV-68) γ Zoonotic infection found in 4.5% of general population and more common in laboratory workers handling infected mice.[14] ELISA tests show factor-of-four (x4) false positive results, due to antibody cross-reaction with other Herpes viruses.
In animal virology the most important herpesviruses belong to the Alphaherpesvirinae. Research on pseudorabies virus (PrV), the causative agent of Aujeszky's disease in pigs, has pioneered animal disease control with genetically modified vaccines. PrV is now extensively studied as a model for basic processes during lytic herpesvirus infection, and for unravelling molecular mechanisms of herpesvirus neurotropism, whereas bovine herpesvirus 1, the causative agent of bovine infectious rhinotracheitis and pustular vulvovaginitis, is analyzed to elucidate molecular mechanisms of latency. The avian infectious laryngotracheitis virus is phylogenetically distant from these two viruses and serves to underline similarity and diversity within the Alphaherpesvirinae.
Subfamily Alphaherpesvirinae
Genus Simplexvirus
Bovine herpesvirus 2 causes bovine mammillitis and pseudo-lumpyskin disease.
Cercopithecine herpesvirus 1, also known as Herpes B virus, causes a Herpes simplex-like disease in Macaques, usually fatal if symptomatic and untreated in humans.
Ateline herpesvirus 1, Spider monkey herpesvirus.
Genus Varicellovirus
Bovine herpesvirus 1 causes infectious bovine rhinotracheitis, vaginitis, balanoposthitis, and abortion in cattle.
Bovine herpesvirus 5 causes encephalitis in cattle.
Caprine herpesvirus 1 causes conjunctivitis and respiratory disease in goats.
Porcine herpesvirus 1 causes pseudorabies.
Equine herpesvirus 1 causes respiratory disease, neurological disease/paralysis, and spontaneous abortion in horses.
Equine herpesvirus 3 causes coital exanthema in horses.
Equine herpesvirus 4 causes rhinopneumonitis in horses.
Canine herpesvirus 1 causes a severe hemorrhagic disease in puppies.
Feline herpesvirus 1 causes feline viral rhinotracheitis and keratitis in cats.
Duck herpesvirus 1 causes duck plague.
Genus Mardivirus
Gallid herpesvirus 2 causes Marek's disease.
Gallid herpesvirus 3 (GaHV-3 or MDV-2)
Herpesvirus of turkeys (HVT)
Genus Iltovirus
Gallid herpesvirus 1 causes infectious laryngotracheitis in birds.
Subfamily Betaherpesvirinae
Porcine herpesvirus 2 causes inclusion body rhinitis in swine.
Subfamily Gammaherpesvirinae
Genus Rhadinovirus
Alcelaphine herpesvirus 1 causes bovine malignant catarrhal fever.
Alcelaphine herpesvirus 2 causes an antelope and hartebeest version of MCF.
Bovine herpesvirus 4
Equine herpesvirus 2 causes equine cytomegalovirus infection.
Equine herpesvirus
Murid herpesvirus 4 Also known as Murine gammaherpesvirus-68 (MHV-68)
[edit] Family Herpesviridae
The following genera are included in the family Herpesviridae:
Subfamily Alphaherpesvirinae
Genus Iltovirus; type species: Gallid herpesvirus 1
Species: Gallid herpesvirus 1, Psittacid herpesvirus 1
Genus Mardivirus; type species: Gallid herpesvirus 2
Species: Columbid herpesvirus 1, Gallid herpesvirus 2, Gallid herpesvirus 3, Meleagrid herpesvirus 1
Genus Simplexvirus; type species: Human herpesvirus 1
Species: Ateline herpesvirus 1, Bovine herpesvirus 2, Cercopithecine herpesvirus 2, Human herpesvirus 1, Human herpesvirus 2, Leporid herpesvirus 4, Macacine herpesvirus 1, Macropodid herpesvirus 1, Macropodid herpesvirus 2, Papiine herpesvirus 2, Saimiriine herpesvirus 1
Genus Unassigned
Species: Chelonid herpesvirus 5, Chelonid herpesvirus 6
Genus Varicellovirus; type species: Human herpesvirus 3
Species: Bovine herpesvirus 1, Bovine herpesvirus 5, Bubaline herpesvirus 1, Canid herpesvirus 1, Caprine herpesvirus 1, Cercopithecine herpesvirus 9, Cervid herpesvirus 1, Cervid herpesvirus 2, Equid herpesvirus 1, Equid herpesvirus 3, Equid herpesvirus 4, Equid herpesvirus 8, Equid herpesvirus 9, Felid herpesvirus 1, Human herpesvirus 3, Phocid herpesvirus 1, Suid herpesvirus
Subfamily Betaherpesvirinae
Genus Cytomegalovirus; type species: Human herpesvirus 5
Species: Cercopithecine herpesvirus 5, Human herpesvirus 5, Macacine herpesvirus 3, Panine herpesvirus 2
Genus Muromegalovirus; type species: Murid herpesvirus 1
Species: Murid herpesvirus 1, Murid herpesvirus 2
Genus Proboscivirus; type species: Elephantid herpesvirus 1
Genus Roseolovirus; type species: Human herpesvirus 6
Species: Human herpesvirus 6, Human herpesvirus 7
Genus Unassigned
Species: Caviid herpesvirus 2, Suid herpesvirus 2, Tupaiid herpesvirus 1
Subfamily Gammaherpesvirinae
Genus Lymphocryptovirus; type species: Human herpesvirus 4
Species: Callitrichine herpesvirus 3, Cercopithecine herpesvirus 14, Gorilline herpesvirus 1, Human herpesvirus 4, Macacine herpesvirus 4, Panine herpesvirus 1, Papiine herpesvirus 1, Pongine herpesvirus 2
Genus Macavirus; type species: Alcelaphine herpesvirus 1
Species: Alcelaphine herpesvirus 1, Alcelaphine herpesvirus 2, Bovine herpesvirus 6, Caprine herpesvirus 2, Hippotragine herpesvirus 1, Ovine herpesvirus 2, Suid herpesvirus 3, Suid herpesvirus 4, Suid herpesvirus 5
Genus Percavirus; type species: Equid herpesvirus 2
Species: Equid herpesvirus 2, Equid herpesvirus 5, Mustelid herpesvirus 1
Genus Rhadinovirus; type species: Saimiriine herpesvirus 2
Species: Ateline herpesvirus 2, Ateline herpesvirus 3, Bovine herpesvirus 4, Human herpesvirus 8, Macacine herpesvirus 5, Murid herpesvirus 4, Saimiriine herpesvirus 2
Genus Unassigned
Species: Equid herpesvirus 7, Phocid herpesvirus 2, Saguinine herpesvirus 1
Subfamily Unassigned
Genus Unassigned
Species: Iguanid herpesvirus 2
Species: duck enteritis virus
Research is currently ongoing into a variety of side-effect or co-conditions related to the herpesviruses. These include:
Alzheimer's disease (This database compiles Alzheimer's disease susceptibility genes related to the Herpes simplex life cycle)
atherosclerosis
cholangiocarcinoma
Crohn's disease
chronic fatigue syndrome
dysautonomia
fibromyalgia
Irritable bowel syndrome
multiple sclerosis
labile hypertension
lupus
pancreatic cancer
pancreatitis
pityriasis rosea
Type II Diabetes
Viral structure
Herpesviruses all share a common structure—all herpesviruses are composed of relatively large double-stranded, linear DNA genomes encoding 100-200 genes encased within an icosahedral protein cage called the capsid which is itself wrapped in a protein layer called the tegument containing both viral proteins and viral mRNAs and a lipid bilayer membrane called the envelope. This whole particle is known as a virion.
Herpes virus life-cycle
All herpesviruses are nuclear-replicating—the viral DNA is transcribed to RNA within the infected cell's nucleus.
Infection is initiated when a viral particle contacts a cell with specific types of receptor molecules on the cell surface. Following binding of viral envelope glycoproteins to cell membrane receptors, the virion is internalized and dismantled, allowing viral DNA to migrate to the cell nucleus. Within the nucleus, replication of viral DNA and transcription of viral genes occurs.
During symptomatic infection, infected cells transcribe lytic viral genes. In some host cells, a small number of viral genes termed latency associated transcript (LAT) accumulate instead. In this fashion the virus can persist in the cell (and thus the host) indefinitely. While primary infection is often accompanied by a self-limited period of clinical illness, long-term latency is symptom-free.
Reactivation of latent viruses has been implicated in a number of diseases (e.g. Shingles, Pityriasis Rosea). Following activation, transcription of viral genes transitions from latency-associated LAT to multiple lytic genes; these lead to enhanced replication and virus production. Often, lytic activation leads to cell death. Clinically, lytic activation is often accompanied by emergence of non-specific symptoms such as low grade fever, headache, sore throat, malaise, and rash as well as clinical signs such as swollen or tender lymph nodes and immunological findings such as reduced levels of natural killer cells.
Taxonomy
The genus Herpesvirus was established in 1971 in the first report of the International Committee on Taxonomy of Viruses (ICTV). This genus consisted of 23 viruses and 4 groups of viruses. In the second ICTV report in 1976 this genus was elevated to family level - the Herpetoviridae. Because of possible confusion with viruses derived from reptiles this name was changed in the third report in 1979 to Herpesviridae. In this report the family Herpesviridae was divided into 3 subfamilies (Alphaherpesvirinae, Betaherpesvirinae and Gammaherpesvirinae) and 5 unnamed genera: 21 viruses were listed. In 2009 the family Herpesviridae was elevated to the order Herpesvirales. This elevation was necessitated by the discovery that the herpes viruses of fish and molluscs were only distantly related to those of birds and mammals. Two new families were created - the family Alloherpesviridae which incorporates bony fish and frog viruses and the family Malacoherpesviridae which contains those of molluscs.
This order currently has 3 families, 3 subfamilies plus 1 unassigned, 17 genera, 90 species and plus 48 as yet unassigned viruses.
Virus naming system
The system of naming herpes viruses was originated in 1973 and has been elaborated considerably since. The recommended naming system specified that each herpes virus should be named after the taxon (family or subfamily) to which its primary natural host belongs. The subfamily name is used for viruses from members of the family Bovidae or from primates (the virus name ending in –ine, e.g. bovine) and the host family name for other viruses (ending in –id, e.g. equid). Human herpes viruses have been treated as an exception (human rather than hominid). Following the host-derived term, the word herpes virus is added, followed by an arabic number (1,2,3,...). These last two additions bear no implied meaning about taxonomic or biological properties of the virus.
Some exceptions to this system exist. A number of viruses' names (e.g. Epstein–Barr virus) are so widely used that it is impractical to attempt to insist on their replacement. This has led to the a dual nomenclature in the literature for some herpes viruses. All herpes viruses described since this system was adopted have been named in accordance with it.
Immune system evasions
cmvIL-10
Herpesviruses are known for their ability to establish lifelong infections. One way this is possible is through immune evasion. Herpesviruses have found many different ways to evade the immune system. One such way is by encoding a protein mimicking human interleukin 10 (hIL-10) and another is by downregulation of the Major Histocompatibility Complex II (MHC II) in infected cells. Research conducted on cytomegalovirus (CMV) indicates that the viral human IL-10 homolog, cmvIL-10, is important in inhibiting pro-inflammatory cytokine synthesis. The cmvIL-10 protein has 27% identity with hIL-10 and only one conserved residue out of the nine amino acids that make up the functional site for cytokine synthesis inhibition on hIL-10. There is, however, much similarity in the functions of hIL-10 and cmvIL-10. Both have been shown to down regulate IFN-γ, IL-1α, GM-CSF, IL-6 and TNF- α, which are all pro-inflammatory cytokines. They have also been shown to play a role in downregulating MHC I and MHC II and up regulating HLA-G (non-classical MHC I). These two events allow for immune evasion by suppressing the cell-mediated immune response and natural killer cell response, respectively. The similarities between hIL-10 and cmvIL-10 may be explained by the fact that hIL-10 and cmvIL-10 both use the same cell surface receptor, the hIL-10 receptor. One difference in the function of hIL-10 and cmvIL-10 is that hIL-10 causes human peripheral blood mononuclear cells (PBMC) to both increase and decrease in proliferation whereas cmvIL-10 only causes a decrease in proliferation of PBMCs. This indicates that cmvIL-10 may lack the stimulatory effects that hIL-10 has on these cells.
It was found that cmvIL-10 functions through phosphorylation of the Stat3 protein. It was originally thought that this phosphorylation was a result of the JAK-STAT pathway. However, despite evidence that JAK does indeed phosphorylate Stat3, its inhibition has no significant influence on cytokine synthesis inhibition. Another protein, PI3K, was also found to phosphorylate Stat3. PI3K inhibition, unlike JAK inhibition, did have a significant impact on cytokine synthesis. The difference between PI3K and JAK in Stat3 phosphorylation is that PI3K phosphorylates Stat3 on the S727 residue whereas JAK phosphorylates Stat3 on the Y705 residue. This difference in phosphorylation positions seems to be the key factor in Stat3 activation leading to inhibition of pro-inflammatory cytokine synthesis. In fact, when a PI3K inhibitor is added to cells, the cytokine synthesis levels are significantly restored. The fact that cytokine levels are not completely restored indicates there is another pathway activated by cmvIL-10 that is inhibiting cytokine synthesis. The proposed mechanism is that cmvIL-10 activates PI3K which in turn activates PKB (Akt). PKB may then activate mTOR, which may target Stat3 for phosphorylation on the S727 residue.[6]
Another one of the many ways in which herpes viruses evade the immune system is by down regulation of MHC I and MHC II. This is observed in almost every human herpesvirus. Down regulation of MHC I and MHC II can come about by many different mechanisms, most causing the MHC to be absent from the cell surface. As discussed above, one way is by a viral chemokine homolog such as IL-10. Another mechanism to down regulate MHCs is to encode viral proteins that detain the newly formed MHC in the endoplasmic reticulum (ER). The MHC cannot reach the cell surface and therefore cannot activate the T cell response. The MHCs can also be targeted for destruction in the proteasome or lysosome. The ER protein TAP also plays a role in MHC down regulation. Viral proteins inhibit TAP preventing the MHC from picking up a viral antigen peptide. This prevents proper folding of the MHC and therefore the MHC does not reach the cell surface.
It is important to note that HLA-G is often up regulated in addition to downregulation of MHC I and MHC II. This prevents the natural killer cell response.[citation needed]
There are eight distinct viruses in this family known to cause disease in humans.[8][9]
Human Herpesvirus (HHV) classification
Type Synonym Subfamily Primary Target Cell Pathophysiology Site of Latency Means of Spread
HHV‑1 Herpes simplex virus-1 (HSV-1) α (Alpha) Mucoepithelial Oral and/or genital herpes (predominantly orofacial), as well as other herpes simplex infections Neuron Close contact (sexually transmitted disease)
HHV-2 Herpes simplex virus-2 (HSV-2) α Mucoepithelial Oral and/or genital herpes (predominantly genital), as well as other herpes simplex infections Neuron Close contact (sexually transmitted disease)
HHV-3 Varicella zoster virus (VZV) α Mucoepithelial Chickenpox and shingles Neuron Respiratory and close contact (including sexually transmitted disease)
HHV-4 Epstein-Barr virus (EBV), lymphocryptovirus γ (Gamma) B cells and epithelial cells Infectious mononucleosis, Burkitt's lymphoma, CNS lymphoma in AIDS patients,
post-transplant lymphoproliferative syndrome (PTLD), nasopharyngeal carcinoma, HIV-associated hairy leukoplakia B cell Close contact, transfusions, tissue transplant, and congenital
HHV-5 Cytomegalovirus (CMV) β (Beta) Monocyte, lymphocyte, and epithelial cells Infectious mononucleosis-like syndrome,[10] retinitis, etc. Monocyte, lymphocyte, and ? Saliva
HHV-6 Roseolovirus, Herpes lymphotropic virus β T cells and ? Sixth disease (roseola infantum or exanthem subitum) T cells and ? Respiratory and close contact?
HHV-7 Roseolovirus β T cells and ? Sixth disease (roseola infantum or exanthem subitum) T cells and ? ?
HHV-8 Kaposi's sarcoma-associated herpesvirus
(KSHV), a type of rhadinovirus γ Lymphocyte and other cells Kaposi's sarcoma, primary effusion lymphoma, some types of multicentric Castleman's disease B cell Close contact (sexual), saliva?
[edit] Herpesviruses of other animals
In addition to the herpesviruses considered endemic in humans, some viruses associated primarily with animals may infect humans. These are zoonotic infections:
Zoonotic Herpesviruses
Species Type Synonym Subfamily Human Pathophysiology
Macaque monkey CeHV-1 Cercopithecine herpesvirus-1, (Monkey B virus) α Very unusual, with only approximately 25 human cases reported.[11] Untreated infection is often deadly; sixteen of the 25 cases resulted in fatal encephalomyelitis. At least four cases resulted in survival with severe neurologic impairment.[11][12] Symptom awareness and early treatment are important for laboratory workers facing exposure.
Mouse MuHV‑4 Murine gammaherpesvirus-68 (MHV-68) γ Zoonotic infection found in 4.5% of general population and more common in laboratory workers handling infected mice.[14] ELISA tests show factor-of-four (x4) false positive results, due to antibody cross-reaction with other Herpes viruses.
In animal virology the most important herpesviruses belong to the Alphaherpesvirinae. Research on pseudorabies virus (PrV), the causative agent of Aujeszky's disease in pigs, has pioneered animal disease control with genetically modified vaccines. PrV is now extensively studied as a model for basic processes during lytic herpesvirus infection, and for unravelling molecular mechanisms of herpesvirus neurotropism, whereas bovine herpesvirus 1, the causative agent of bovine infectious rhinotracheitis and pustular vulvovaginitis, is analyzed to elucidate molecular mechanisms of latency. The avian infectious laryngotracheitis virus is phylogenetically distant from these two viruses and serves to underline similarity and diversity within the Alphaherpesvirinae.
Subfamily Alphaherpesvirinae
Genus Simplexvirus
Bovine herpesvirus 2 causes bovine mammillitis and pseudo-lumpyskin disease.
Cercopithecine herpesvirus 1, also known as Herpes B virus, causes a Herpes simplex-like disease in Macaques, usually fatal if symptomatic and untreated in humans.
Ateline herpesvirus 1, Spider monkey herpesvirus.
Genus Varicellovirus
Bovine herpesvirus 1 causes infectious bovine rhinotracheitis, vaginitis, balanoposthitis, and abortion in cattle.
Bovine herpesvirus 5 causes encephalitis in cattle.
Caprine herpesvirus 1 causes conjunctivitis and respiratory disease in goats.
Porcine herpesvirus 1 causes pseudorabies.
Equine herpesvirus 1 causes respiratory disease, neurological disease/paralysis, and spontaneous abortion in horses.
Equine herpesvirus 3 causes coital exanthema in horses.
Equine herpesvirus 4 causes rhinopneumonitis in horses.
Canine herpesvirus 1 causes a severe hemorrhagic disease in puppies.
Feline herpesvirus 1 causes feline viral rhinotracheitis and keratitis in cats.
Duck herpesvirus 1 causes duck plague.
Genus Mardivirus
Gallid herpesvirus 2 causes Marek's disease.
Gallid herpesvirus 3 (GaHV-3 or MDV-2)
Herpesvirus of turkeys (HVT)
Genus Iltovirus
Gallid herpesvirus 1 causes infectious laryngotracheitis in birds.
Subfamily Betaherpesvirinae
Porcine herpesvirus 2 causes inclusion body rhinitis in swine.
Subfamily Gammaherpesvirinae
Genus Rhadinovirus
Alcelaphine herpesvirus 1 causes bovine malignant catarrhal fever.
Alcelaphine herpesvirus 2 causes an antelope and hartebeest version of MCF.
Bovine herpesvirus 4
Equine herpesvirus 2 causes equine cytomegalovirus infection.
Equine herpesvirus
Murid herpesvirus 4 Also known as Murine gammaherpesvirus-68 (MHV-68)
[edit] Family Herpesviridae
The following genera are included in the family Herpesviridae:
Subfamily Alphaherpesvirinae
Genus Iltovirus; type species: Gallid herpesvirus 1
Species: Gallid herpesvirus 1, Psittacid herpesvirus 1
Genus Mardivirus; type species: Gallid herpesvirus 2
Species: Columbid herpesvirus 1, Gallid herpesvirus 2, Gallid herpesvirus 3, Meleagrid herpesvirus 1
Genus Simplexvirus; type species: Human herpesvirus 1
Species: Ateline herpesvirus 1, Bovine herpesvirus 2, Cercopithecine herpesvirus 2, Human herpesvirus 1, Human herpesvirus 2, Leporid herpesvirus 4, Macacine herpesvirus 1, Macropodid herpesvirus 1, Macropodid herpesvirus 2, Papiine herpesvirus 2, Saimiriine herpesvirus 1
Genus Unassigned
Species: Chelonid herpesvirus 5, Chelonid herpesvirus 6
Genus Varicellovirus; type species: Human herpesvirus 3
Species: Bovine herpesvirus 1, Bovine herpesvirus 5, Bubaline herpesvirus 1, Canid herpesvirus 1, Caprine herpesvirus 1, Cercopithecine herpesvirus 9, Cervid herpesvirus 1, Cervid herpesvirus 2, Equid herpesvirus 1, Equid herpesvirus 3, Equid herpesvirus 4, Equid herpesvirus 8, Equid herpesvirus 9, Felid herpesvirus 1, Human herpesvirus 3, Phocid herpesvirus 1, Suid herpesvirus
Subfamily Betaherpesvirinae
Genus Cytomegalovirus; type species: Human herpesvirus 5
Species: Cercopithecine herpesvirus 5, Human herpesvirus 5, Macacine herpesvirus 3, Panine herpesvirus 2
Genus Muromegalovirus; type species: Murid herpesvirus 1
Species: Murid herpesvirus 1, Murid herpesvirus 2
Genus Proboscivirus; type species: Elephantid herpesvirus 1
Genus Roseolovirus; type species: Human herpesvirus 6
Species: Human herpesvirus 6, Human herpesvirus 7
Genus Unassigned
Species: Caviid herpesvirus 2, Suid herpesvirus 2, Tupaiid herpesvirus 1
Subfamily Gammaherpesvirinae
Genus Lymphocryptovirus; type species: Human herpesvirus 4
Species: Callitrichine herpesvirus 3, Cercopithecine herpesvirus 14, Gorilline herpesvirus 1, Human herpesvirus 4, Macacine herpesvirus 4, Panine herpesvirus 1, Papiine herpesvirus 1, Pongine herpesvirus 2
Genus Macavirus; type species: Alcelaphine herpesvirus 1
Species: Alcelaphine herpesvirus 1, Alcelaphine herpesvirus 2, Bovine herpesvirus 6, Caprine herpesvirus 2, Hippotragine herpesvirus 1, Ovine herpesvirus 2, Suid herpesvirus 3, Suid herpesvirus 4, Suid herpesvirus 5
Genus Percavirus; type species: Equid herpesvirus 2
Species: Equid herpesvirus 2, Equid herpesvirus 5, Mustelid herpesvirus 1
Genus Rhadinovirus; type species: Saimiriine herpesvirus 2
Species: Ateline herpesvirus 2, Ateline herpesvirus 3, Bovine herpesvirus 4, Human herpesvirus 8, Macacine herpesvirus 5, Murid herpesvirus 4, Saimiriine herpesvirus 2
Genus Unassigned
Species: Equid herpesvirus 7, Phocid herpesvirus 2, Saguinine herpesvirus 1
Subfamily Unassigned
Genus Unassigned
Species: Iguanid herpesvirus 2
Species: duck enteritis virus
Research is currently ongoing into a variety of side-effect or co-conditions related to the herpesviruses. These include:
Alzheimer's disease (This database compiles Alzheimer's disease susceptibility genes related to the Herpes simplex life cycle)
atherosclerosis
cholangiocarcinoma
Crohn's disease
chronic fatigue syndrome
dysautonomia
fibromyalgia
Irritable bowel syndrome
multiple sclerosis
labile hypertension
lupus
pancreatic cancer
pancreatitis
pityriasis rosea
Type II Diabetes













1 komentar:
I want to share my testimonies to the general
public on how this great man called Dr OLOKUN who cure my herpes. I have been a HERPES patient for over 9 Months and i have been in pains until i came across this lady when i traveled to Africa for business trip who happer and she told me that there is this great herbal doctor that help her tocure her herpes and she gave me his email address for me to contact and i did as she instructed. The man Dr OLOKUN told me how much to buy the
herpes herbal medication and how i will get it, which i did. And to my
greatest surprise that i took the Herpes herbal medicine for just two week as he
instructed and behold i went for a herpes test, to my greatest surprise for the
doctor confirmed me to be herpes free and said that i no longer have
herpes in my system and till now i have never felt any pains nor herpes again, so i
said i must testify the goodness of this man to the general public. So if
you are there surfing from this Herpes problems or any deadly disease or
other disease for i will advice you to contact him on his working
email: drolokuntemple@gmail.com whatsapp him with thise number +2348132537313
Posting Komentar